New antipsychotic blood test reduces wait for results from ten days to six minutes

ReStart and South London and Maudsley NHS Trust (SLaM) have joined forces to pioneer a new antipsychotic blood testing process for patients which delivers results within minutes.
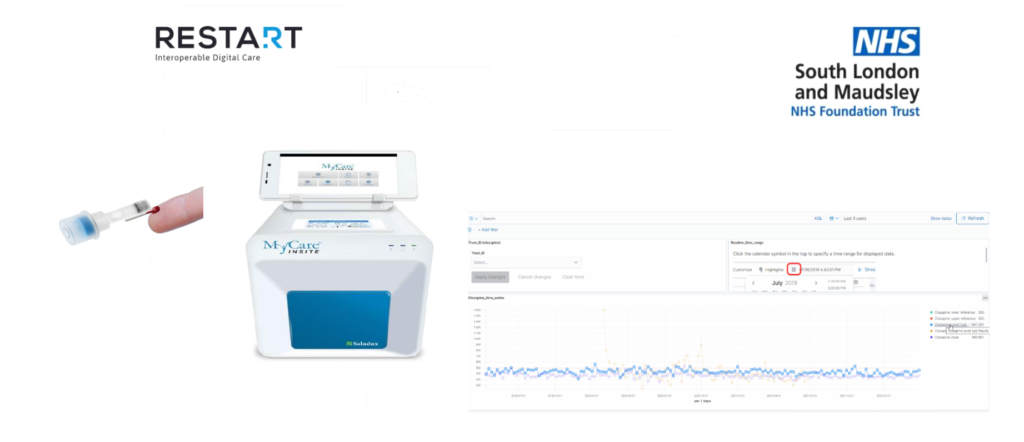
The new finger prick test, provided by Saladax Biomedical, reduces the need for traditional blood testing, cutting the time spent waiting for results from 5-10 days to around 6 minutes.
SLaM have collaborated with integration and interoperability provider ReStart to connect systems across the whole South East London ICS and the wider Local Health and Care Record Exemplar, One London.
This integration means that results data can be moved quickly between collection devices, allowing them to be displayed in real time. This supports rapid clinical decision-making regarding dose adjustments based on side effects and symptoms. The new process saves time and resources, with the potential to reduce inpatient bed stays to 3 weeks, instead of 3 months.
Andy Meiner, Chief Revenue Officer, for ReStart said: “We are proud to be the interoperability provider to SLaM NHS Trust. They remain the mental health experts within the NHS, spearheading ground breaking research which in turn improves patient outcomes, saves resources and shortens care journeys.”
Having instantaneous results at the point of care means that patients are better informed and demonstrate increased adherence and engagement with their treatment. This has led to improved patient outcomes.
The work utilises TIE interoperability across data collection points, patient records and Cogstack AI for the processing of results.
Stuart MacLellan, Chief Information Officer at SLaM, said: “This new blood testing system has the potential to significantly reduce the length of inpatient stays. This is invaluable given the significant demand for NHS mental health services now.
“Not only does this test reduce pressure on services, it empowers patients, giving them more control around their care – ultimately improving outcomes. We look forward to seeing the positive impact this can have across the NHS, as it continues to benefit our organisation.
“This wouldn’t have been possible without interoperability provider ReStart, allowing results data to be seen in real time and supporting rapid clinical decision making.”
Sal Salamone, Founder and CEO of Saladax Biomedical, said: “The ability to partner with an outstanding organisation such as South London and Maudsley NHS Trust to implement our PoC device was an incredible opportunity to fulfil the vision for our company.
“From the improved patient-friendly blood collection to providing immediate test results while the patient is present, will significantly improve patient care and the drug’s effectiveness. In addition, this disruptive technology will dramatically impact the use and efficacy of clozapine drug treatment.”
To aid this work, the SLaM team have created a visualisation display for the outputs of the PoC test results, which gives clinicians direct access to results and the ability to calculate trends across defined time periods.
The published studies that have been carried out to date, show considerable benefits in financial, planning and patient outcomes related to this new test technique. Under the new testing and medication regime, patients can be stabilised on clozapine at a much shorter time, saving thousands and improving patients’ lives.
